Ameloot Group
Manipulating Porous Matter
Papers
Vapor-assisted synthesis of the MOF-74 family @Dalton Trans.!
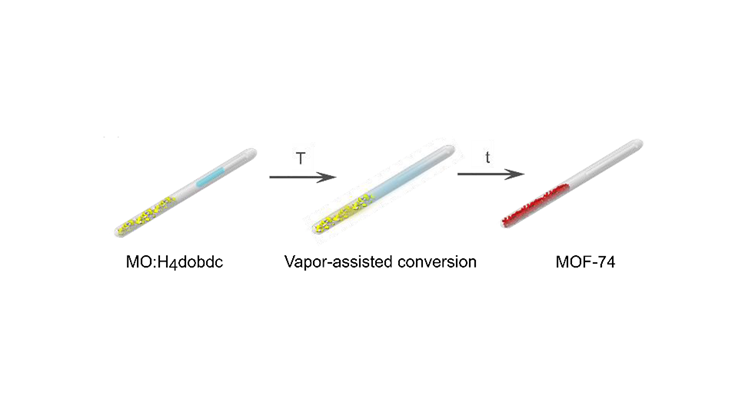
In this work, we investigate the vapor-assisted synthesis of the metal-organic framework MOF-74 starting from three metal oxides (ZnO, CoO, and MgO). Depending on the nature of the added vapor (H2O, DMF, DMSO), the metal oxide, and the temperature, the outcome of the reaction can be directed towards the desired porous phase.
Ex situ and in situ XRD measurements reveal the formation of an intermediate phase during the reaction of MgO with H4dobdc, while the MOF-74 phase forms directly for ZnO and CoO. The reduced CO2 uptake of the resulting materials compared to solvothermally prepared MOFs might be offset by the convenience of the presented route and the promise of a high space time yield.
More information van be found here, or on our publication page.
Digital halftoning for printer-independent SLA of functionally graded materials @Cell Rep. Phys. Sci.!
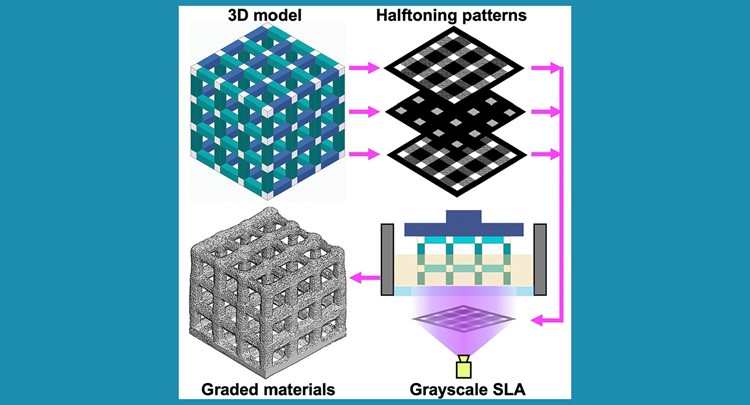
Grayscale masks have been applied in high-end stereolithography (SLA) 3D printers to spatially define the degree of photopolymerization and fabricate objects with graded properties. However, this approach cannot be applied in many commonly used projection SLA printers that can only handle 1-bit slices.
Based on dithering techniques and image-processing algorithms, we developed a digital halftoning approach to generate universally printable files consisting only of black and white pixels arranged to mimic the ‘‘true’’ grayscale levels. Using this approach, any SLA printer can be used to control the irradiation dose and the degree of photopolymerization in each printed layer. As proof of concept, we report various objects with graded properties using different commercial printers. This universal fabrication strategy will open a new window for engineers and scientists to explore grayscale SLA prints.
More information can be found here, or on the publication page.
Thin film capacitor geometries for VOC detection @ ACS Sensors!
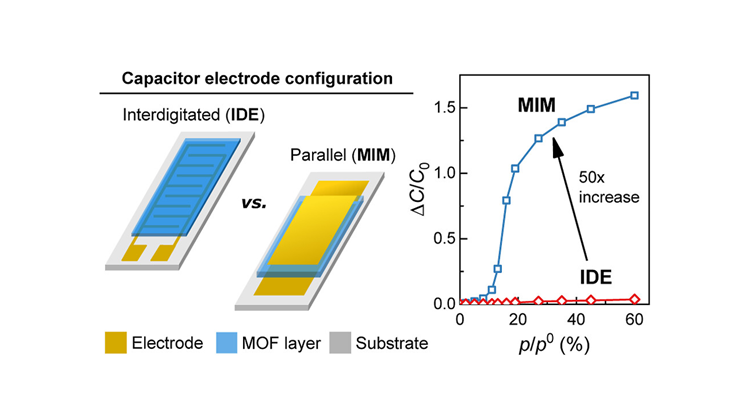
Their chemical diversity, uniform pore sizes, and large internal surface areas make metal−organic frameworks (MOFs) highly suitable for volatile organic compound (VOC) adsorption. In this work, we compare two geometries of capacitive VOC sensors that use the MOF material ZIF-8 as an affinity layer. When using a permeable top electrode (thickness < 25 nm), the metal−insulator−metal sandwich configuration exhibits superior sensitivity, an improved detection limit, and a smaller footprint than the conventional interdigitated electrode layout. Moreover, the transduction of VOC adsorption in ZIF-8 via MIM capacitors is more sensitive to polar VOCs and provides better selectivity at high loadings than gravimetric and optical transductions.
More information can be found here, or on the publication page of our website.
SLA 3D printing of graded porous materials @Cell Reports Physical Science!
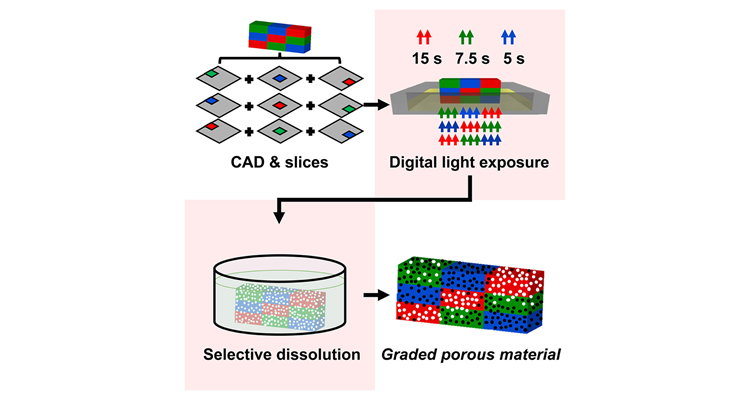
Graded porous materials (GPMs) with fine pore sizes (<50 μm) and gradient porosity are used in diverse fields such as biomedical implants, sensors, and soft robotics. Stereolithography (SLA) 3D printing has created new possibilities for fabricating intricate porous materials due to its high speed and excellent resolution. Nevertheless, for many commonly used low-cost SLA printers, the traditional SLA process is not ideally suited to print GPMs since only a single material feedstock is used for the entire print.
In this paper, we demonstrate how this limitation can be overcome by using a composite feedstock consisting of a photoresin and a polymeric filler in combination with voxel-level digital control over the light exposure. Based on this strategy, spatial control over the porosity, mechanical properties, and swelling behavior within the SLA prints is achieved. We expect that this scalable fabrication strategy will contribute to a variety of applications that require GPMs.
More information can be found here, or on the Publication page.
Ceramic foams through 3D-printed templates @Adv. Eng. Mater.!
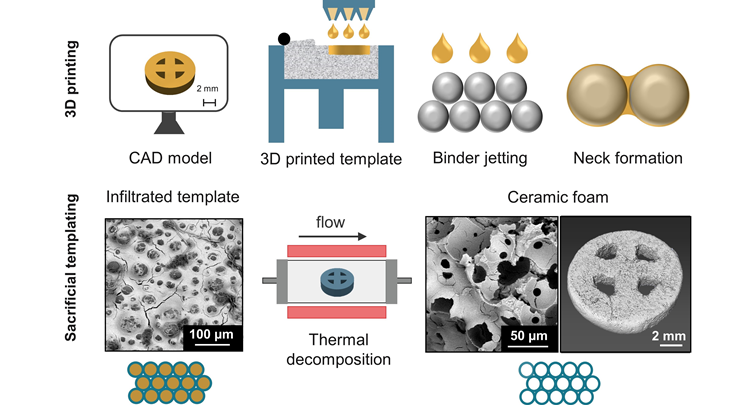
In this paper, powder-based three-dimensional (3D) printing was combined with sacrificial templating to realize highly porous yttria-stabilized ZrO2 (YSZ) ceramic foam objects with well-defined geometries. The porous sacrificial template is 3D-printed using poly(methyl methacrylate) powder. Various methods are evaluated to optimize ceramic slurry infiltration into the 3D-printed template and subsequent burn-out. The optimized method yields ceramic foam objects with an open porosity of > 66% and replicates the geometry of the 3D-printed template with high fidelity.
More information here, or on the publication page!
Molecular layer deposition of MOFs @Chem. of Mat.!
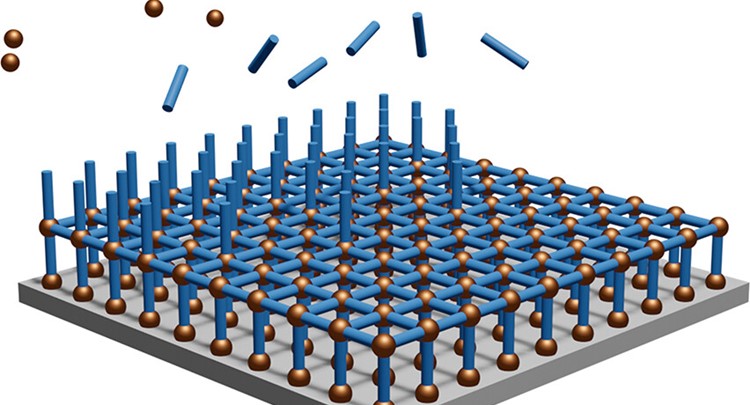
Vapor-phase film deposition of metal–organic frameworks (MOFs) would facilitate the integration of these materials into electronic devices. In this study, we report the vapor-phase layer-by-layer deposition of zeolitic imidazolate framework 8 (ZIF-8) by consecutive, self-saturating reactions of diethyl zinc, water, and 2-methylimidazole on a substrate.
Two approaches were compared: (1) Direct ZIF-8 “molecular layer deposition” (MLD), which enables a nanometer-resolution thickness control and employs only self-saturating reactions, resulting in smooth films that are crystalline as-deposited, and (2) two-step ZIF-8 MLD, in which crystallization occurs during a postdeposition treatment with additional linker vapor. The latter approach resulted in a reduced deposition time and an improved MOF quality, i.e., increased crystallinity and probe molecule uptake, although the smoothness and thickness control were partially lost. Both approaches were developed in a modified atomic layer deposition reactor to ensure cleanroom compatibility.
More information can be found here, or on the Publication page of our website
CVD and H-R patterning of highly conductive 2D coordination polymer film @JACS!
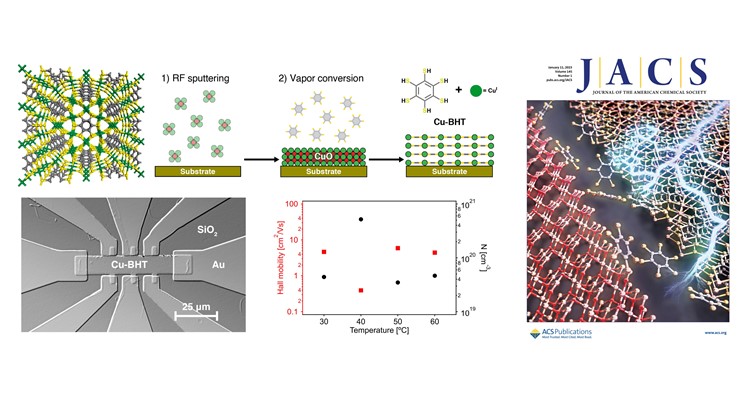
Crystalline coordination polymers with high electrical conductivities and charge carrier mobilities might open new opportunities for electronic devices. However, current solvent-based synthesis methods hinder compatibility with microfabrication standards.
In this report, we describe a solvent-free chemical vapor deposition method to prepare high-quality films of the two-dimensional conjugated coordination polymer Cu-BHT. This approach involves the conversion of a metal oxide precursor into Cu-BHT nanofilms with a controllable thickness (20–85 nm) and low roughness (<10 nm) through exposure to the vaporized organic linker. Moreover, the restricted metal ion mobility during the vapor–solid reaction enables high-resolution patterning via both bottom-up lithography, including the fabrication of micron-sized Hall bar and electrode patterns to accurately evaluate the conductivity and mobility values of the Cu-BHT films.
Check out a visualization of this article on JACS cover!
The article can be accessed here, or via the publication page of our website.
Tunable luminescence from polycyclic aromatic hydrocarbons confined in ZIFs @Adv. Opt. Mat.!
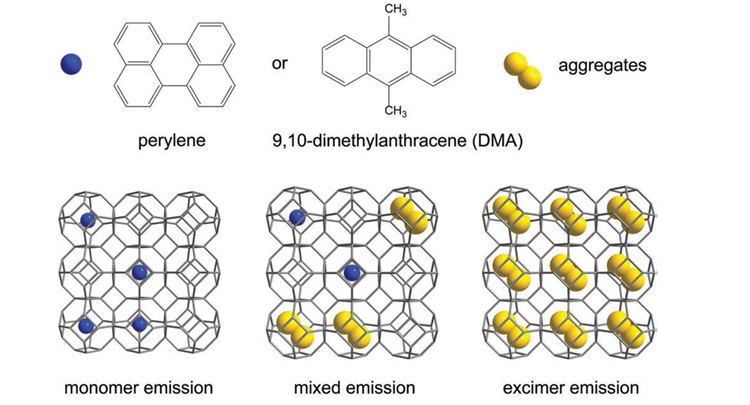
Metal–organic frameworks (MOFs) can provide a variety of nanocompartments for the confinement of guest molecules. Furthermore, the emissions of fluorescent molecules can be tuned by confinement. In this study, a solvent-free “bottle-around-ship” method is used to encapsulate perylene and 9,10-dimethylanthracene, two polycyclic aromatic hydrocarbons, in the MOF ZIF-8. Luminescence color tuning is achieved, including white-light emission, when controlling the loading of only a single type of guest. Photophysical analysis suggests that the variations in luminescence result from various guest arrangements in the nanocompartments, as well as host–guest interactions. Because of the tight confinement of the guests, this host–guest system displays excellent luminescence thermal stability.
More information can be found here, or on the publication page of our website!
Vapor-assisted powder synthesis and oriented MOF-CVD thin films of HKUST-1 @Inorg. Chem.!
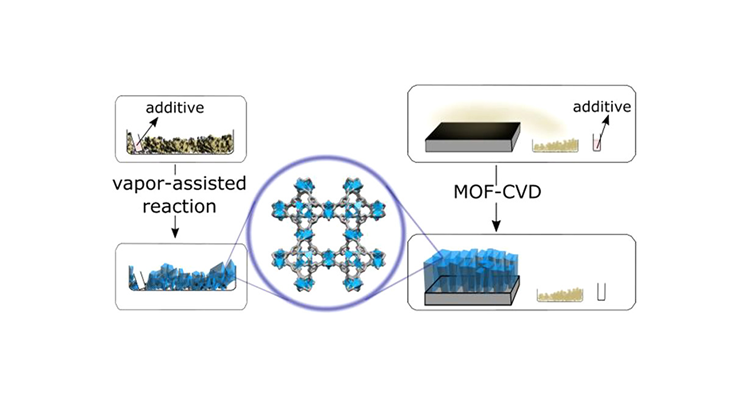
In this paper, we report the development of a vapor-assisted synthesis method for the metal–organic framework (MOF) HKUST-1 in both powder and film format. The use of a solvent template supplied from the vapor phase is essential to form the framework under these conditions. Chemical vapor deposition of HKUST-1 films (MOF-CVD) results in smooth films that show the expected adsorption behavior. Interestingly, the HKUST-1 films obtained this way show a (111) preferred crystallographic orientation.
More information here, or on the Publication page of our website!
Vapour-phase loading of ionic liquids into ZIFs @Inorg. Chem.!
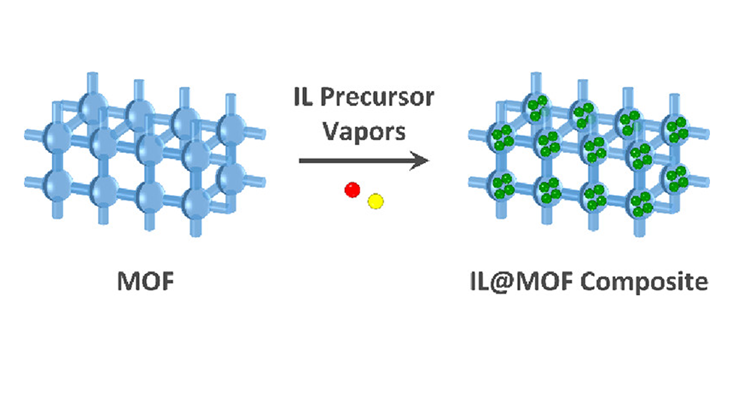
Composites formed by a metal–organic framework (MOF) and an ionic liquid (IL) are potentially interesting materials for applications ranging from gas separation to electrochemical devices. Consequently, there is a need for robust and low-cost preparation procedures that are compatible with the desired applications.
In this paper, we report a solvent-free, one-step, and vapor-based ship-in-bottle synthesis of the IL@MOF composite 1-butyl-3-methylimidazolium bromide@ZIF-8 in powder and thin film forms. In this approach, volatile IL precursors evaporate and subsequently adsorb and react within the MOF cages to form the IL.
More information here or on the Publication page of our website.